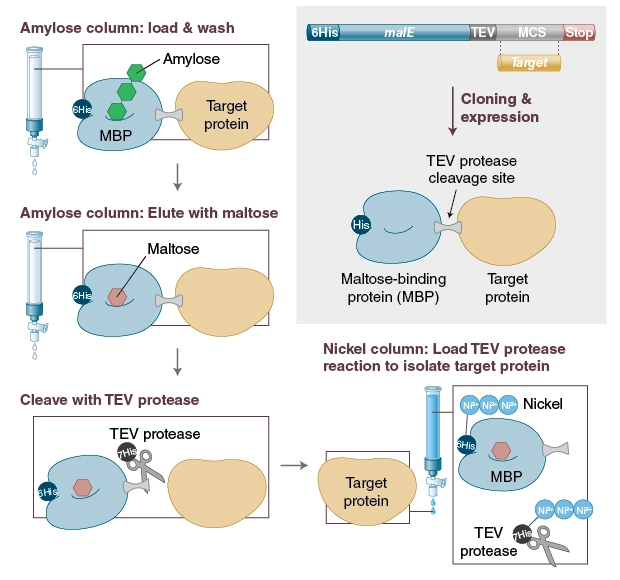
Expression of human ACE2 N-terminal domain, part of the receptor for SARS-CoV-2, in fusion with maltose binding protein, E. coli ribonuclease I and human RNase A | bioRxiv

Recombinant Mbp Mcp Conjugated Amylose Resin (New England Biolabs) | Bioz | Ratings For Life-Science Research

PALI1 facilitates DNA and nucleosome binding by PRC2 and triggers an allosteric activation of catalysis | Nature Communications

Enhanced expression and purification of nucleotide-specific ribonucleases MC1 and Cusativin - ScienceDirect






![NEB] NEBExpress® T4 Lysozyme | 코람바이오텍(주)/코람바이오젠(주) NEB] NEBExpress® T4 Lysozyme | 코람바이오텍(주)/코람바이오젠(주)](https://www.korambiotech.com/wp-content/uploads/2023/06/nebexpress-figure1-min.png)


![NEB] NEBExpress® T4 Lysozyme | 코람바이오텍(주)/코람바이오젠(주) NEB] NEBExpress® T4 Lysozyme | 코람바이오텍(주)/코람바이오젠(주)](https://www.korambiotech.com/wp-content/uploads/2023/06/nebexpress-figure2-min.png)


![NEB] MBP binding protein 으로 solubility 와 expression level을 함께 향상시키세요. | 연구용제품 > Bio마켓 | BRIC NEB] MBP binding protein 으로 solubility 와 expression level을 함께 향상시키세요. | 연구용제품 > Bio마켓 | BRIC](https://www.ibric.org/upload/board/files/new_protech/new_7/photo_0318797.jpg)